Expert CDMO Support for Your Molecule
Comprehensive, complete lifecycle solutions for product development and manufacturing

Power Your Innovations With Our CDMO Services


Process and Analytical Development
We leverage our expertise and advanced capabilities in process and analytical development to ensure seamless transitions from R&D to scalable, high-quality manufacturing solutions.

cGMP Manufacturing
Our versatile facilities and adaptable infrastructure are equipped to support your journey from clinical to commercial manufacturing and meet the demands of each phase in your product’s lifecycle.



Custom Synthesis
Tailored synthesis solutions for organic compounds to meet the unique demands of our partners in the pharmaceutical, electronics, and specialty chemicals industries.
End-to-End Solutions














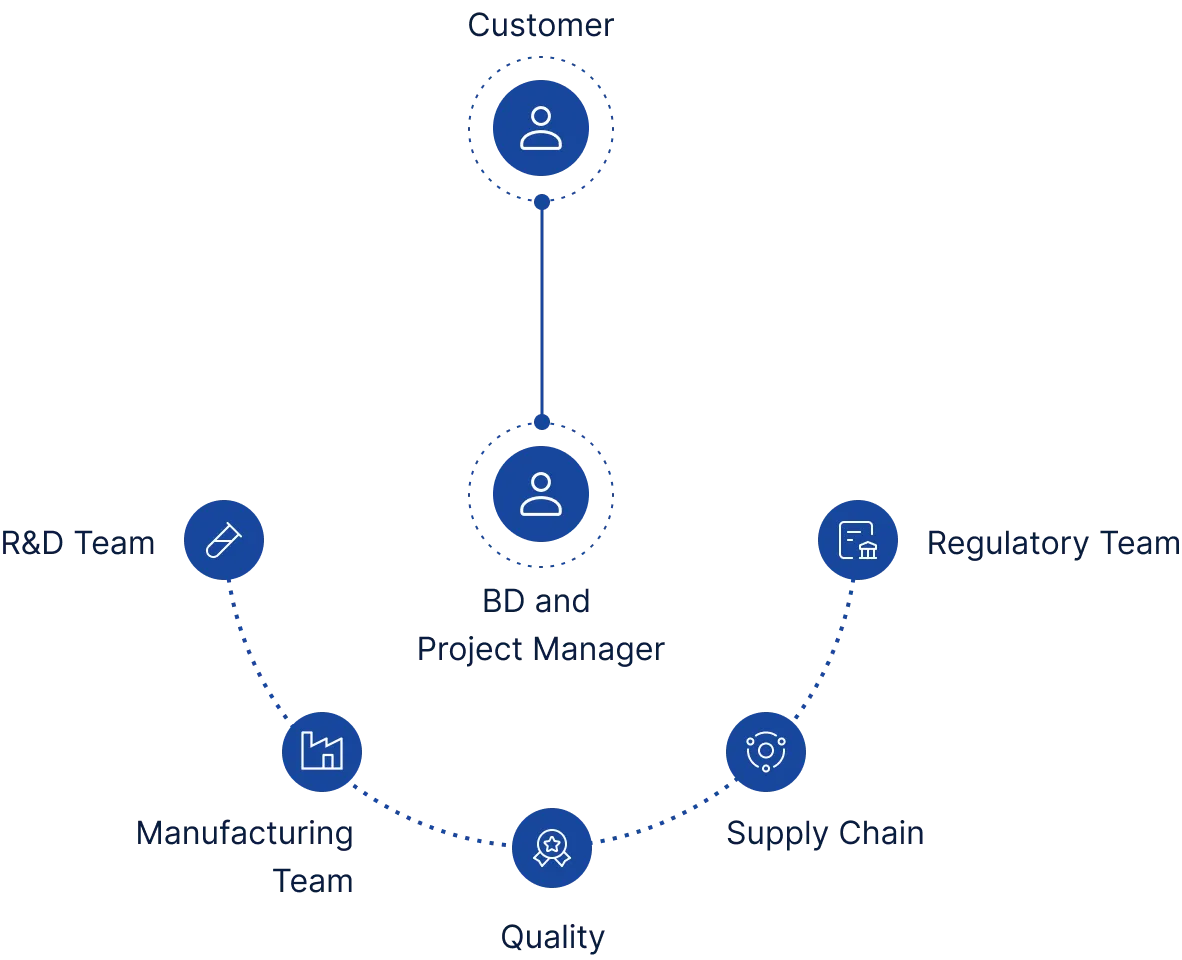
Our customer-centric approach
Our project management strategy holds our customers at its very center – we prioritize transparent communication and cross-functional collaboration for reliable outcomes at every project stage.
Our clients have a dedicated Project Manager who coordinate with key teams to keep them informed and deliver results that meet their goals, every step of the way.